|
In 1963, a boy from Tanzania had found that hot water freeze faster than cold water. Yes, you read right and I'm not drunk. Get the lowdown on "Mpemba effect". (Something like Leidenforst ?) Who is Mpemba and How did he find this effect ? "The effect is named after Tanzanian Erasto Mpemba. He described it in 1963 in Form 3 of Magamba Secondary School, Tanganyika, when freezing ice cream mix that was hot in cookery classes and noticing that it froze before the cold mix. He later became a student at Mkwawa Secondary (formerly High) School in Iringa. The headmaster invited Dr. Denis G. Osborne from the University College in Dar es Salaam to give a lecture on physics. After the lecture, Erasto Mpemba asked him the question "If you take two similar containers with equal volumes of water, one at 35 °C (95 °F) and the other at 100 °C (212 °F), and put them into a freezer, the one that started at 100 °C (212 °F) freezes first. Why?", only to be ridiculed by his classmates and teacher. After initial consternation, Osborne experimented on the issue back at his workplace and confirmed Mpemba's finding. They published the results together in 1969, while Mpemba was studying at the College of African Wildlife Management." It is extracurricular for formal education and can not explain at first sight. Over the years many scientists have puzzled over a counter-intuitive observation: hot water, for some reason, seems to freeze faster than cold water. This phenomenon was explained by a Tanzanian Student, Mpemba when he observed that his warm ice cream mix froze faster than the cold one. Later the team of physicist led by Xi Zhang found that the reason behind this effect is due to the covalent bond present in water. Each molecule of water is made up of two hydrogen atoms bonded covalently to a single atom of oxygen. Those bonds, which involve atoms sharing electrons, are well understood. As shown in figure below:- But the separate water molecules are bound together, too, by weaker forces generated by hydrogen bonds. They occur when a hydrogen atom from one molecule of water sits close to an oxygen atom from another—and they give rise to many of water's interesting properties, like its strangely high boiling point. Those same bonds cause the Mpemba effect. The idea is pretty simple: bring water molecules into close contact, and a natural repulsion between the molecules causes the covalent bonds to stretch and store energy. As the the liquid warms up, the hydrogen bonds stretch as the water gets less dense and the molecules move further apart. That extra stretch in hydrogen bonds allows the covalent bonds to relax and shrink a little, giving up their energy. The process of covalent bonds giving up energy is equivalent to cooling, so warm water should in theory cool faster than cold. This is what the Mpemba effect is! But this effect is not valid in all conditions. Say for example you take two containers of water, which are identical in shape, and which hold identical amounts of water. The only difference between the two is that the water in one is at a higher (uniform) temperature than the water in the other. Now we cool both containers, using the exact same cooling process for each container. Under some conditions the initially warmer water will freeze first. If this occurs, we have seen the Mpemba effect. Of course, the initially warmer water will not freeze before the initially cooler water for all initial conditions. If the hot water starts at 99.9°C, and the cold water at 0.01°C, then clearly under those circumstances, the initially cooler water will freeze first. However, under some conditions the initially warmer water will freeze first: if that happens, you have seen the Mpemba effect. But you will not see the Mpemba effect for just any initial temperatures, container shapes, or cooling conditions. This seems impossible, right? Many sharp readers may have already come up with a common proof that the Mpemba effect is impossible. The proof usually goes something like this. Say that the initially cooler water starts at 30°C and takes 10 minutes to freeze, while the initially warmer water starts out at 70°C. Now the initially warmer water has to spend some time cooling to get to get down to 30°C, and after that, it's going to take 10 more minutes to freeze. So since the initially warmer water has to do everything that the initially cooler water has to do, plus a little more, it will take at least a little longer, right? What can be wrong with this proof? What's wrong with this proof is that it implicitly assumes that the water is characterized solely by a single number — its average temperature. But if other factors besides the average temperature are important, then when the initially warmer water has cooled to an average temperature of 30°C, it may look very different than the initially cooler water (at a uniform 30°C) did at the start. Why? The reason may be because the water may have changed when it cooled down from a uniform 70°C to an average 30°C. It could have less mass, less dissolved gas, or convection currents producing a non-uniform temperature distribution. Or it could have changed the environment around the container in the refrigerator. And in fact the Mpemba effect has been observed in a number of controlled experiments. Mpemba effect depends on some factors which are explained as follows:-
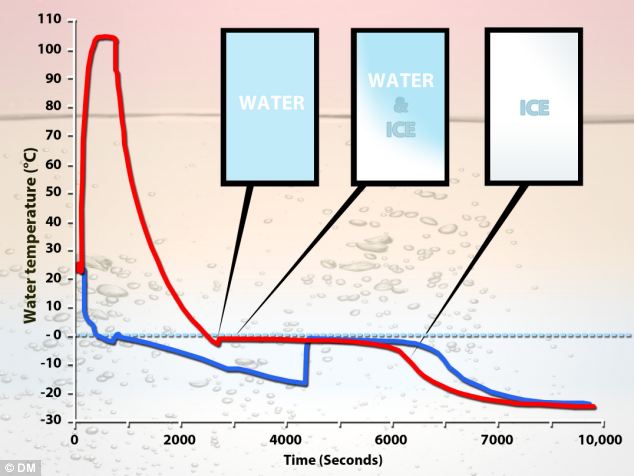
In short, hot water does freeze sooner than cold water under a wide range of circumstances. It is not impossible, and has been seen to occur in a number of experiments. However, despite claims often made by one source or another, there is no well-agreed explanation for how this phenomenon occurs. Different mechanisms have been proposed, but the experimental evidence is inconclusive. "This graph shows how the Mpemba effect - how hot water freezes faster than cold. It shows how rapidly boiling water cools compared to the water that starts off at a lower temperature. Scientists think they have finally solved the mystery and believe the secret lies in the unique properties of the bonds that hold water together."
Sources: [1]http://gizmodo.com/weve-finally-figured-out-why-hot-water-freezes-faster-1455906029 [2]http://math.ucr.edu/home/baez/physics/General/hot_water.html [3]https://phys.org/news/2010-03-mpemba-effect-hot-faster-cold.html [4]http://www.dailymail.co.uk/sciencetech/article-2483383/Mystery-hot-water-freezes-faster-cold-solved--strange-behaviour-atom-bonds.html
0 Comments
|
Categories
All
|