|
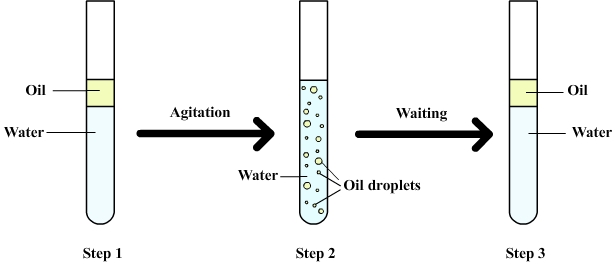
Both water and oil, whether directly or not, the most consumed substances as to sustain daily life. Nonetheless, the mixture of oil and water solution is not homogeneous due to their distinguished molecular structures. I remember that I had had a great effort for the purpose of obtain a homogeneous oil & water mixture when I was just a curious kid. Here a simple -nonscientific-explanation by Ted-Ed why obtaining a homogeneous solution is impossible. It is indeed funny, pursuit of the curiosity: Technically speaking, water and oil molecules differ regarding chemical bound among atoms. Molecules of water are strongly attracted to each other because they are polar. Oil and water molecules are not attracted to each other because oil molecules are non-polar and hydrophobic. Oil has a lower density than water, so it always floats on top. Besides, whilst oil is spilled into the water, the oil molecules immediately try to increase through high level and keep up to float since the density of oil is lower than water. How does the detergent drop out dirty molecules such as oil from fabrics? Detergent molecules are attracted to both water and oil. When you add the detergent, one end of each a detergent molecule attaches to a water molecule and the other attaches to an oil molecule. The detergent creates a mixture of water with droplets of oil spread through it. Detergent is attracted to both oil and water because one end is hydrophilic and sticks to water and the other end is hydrophobic and sticks to oil. When we add detergent to oil and water and shake the mixture, the liquids are held to each other by the detergent molecules and form an emulsion. An emulsion is a stable mixture of two or more liquids that would not normally mix. We use detergent when washing up because it attaches to the oil and grease on dirty dishes and lifts off into the water. Oil also helps animals that live in oceans stay warm because the oil on their feathers or coat keeps the cold water away from their skin [source]. In the literature, there are several methods to investigate multiphase flows. After those examination above, I, of course, have conducted a CFD analysis which encompasses transient sunflower oil spilling process into the water at rest. The analysis is carried out under the conditions below:
I hope you would enjoy with the video. I really fond of grinding.
0 Comments
|
Categories
All
|